Frova Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
FROVA should only be used where a clear diagnosis of migraine has been established.
Risk of Myocardial Ischemia and/or Infarction and Other Adverse Cardiac Events:
Because of the potential of this class of compound (5-HT1 agonists) to cause coronary vasospasm, frovatriptan should not be given to patients with documented ischemic or vasospastic coronary artery disease (CAD) (see CONTRAINDICATIONS). It is strongly recommended that frovatriptan not be given to patients in whom unrecognized CAD is predicted by the presence of risk factors (e.g., hypertension, hypercholesterolemia, smoker, obesity, diabetes, strong family history of CAD, female with surgical or physiological menopause, or male over 40 years of age) unless a cardiovascular evaluation provides satisfactory clinical evidence that the patient is reasonably free of coronary artery and ischemic myocardial disease or other significant underlying cardiovascular disease. The sensitivity of cardiac diagnostic procedures to detect cardiovascular disease or predisposition to coronary artery vasospasm is modest, at best. If, during the cardiovascular evaluation, the patient’s medical history, electrocardiographic, or other investigations reveal findings indicative of, or consistent with, coronary artery vasospasm or myocardial ischemia, frovatriptan should not be administered (see CONTRAINDICATIONS).
For patients with risk factors predictive of CAD, who are determined to have a satisfactory cardiovascular evaluation, it is strongly recommended that administration of the first dose of frovatriptan take place in the setting of a physician’s office or similar medically staffed and equipped facility unless the patient has previously received frovatriptan. Because cardiac ischemia can occur in the absence of clinical symptoms, consideration should be given to obtaining on the first occasion of use an electrocardiogram (ECG) during the interval immediately following administration of FROVA in these patients with risk factors.
It is recommended that patients who are intermittent long-term users of 5-HT1 agonists, including FROVA and who have or acquire risk factors predictive of CAD, as described above, undergo periodic cardiovascular evaluation as they continue to use FROVA.
The systematic approach described above is intended to reduce the likelihood that patients with unrecognized cardiovascular disease would be inadvertently exposed to frovatriptan.
Cardiac Events and Fatalities with 5-HT1 Agonists:
Serious adverse cardiac events, including acute myocardial infarction, life-threatening disturbances of cardiac rhythm and death have been reported within a few hours of administration of 5-HT1 agonists. Considering the extent of use of 5-HT1 agonists in patients with migraine, the incidence of these events is extremely low.
Premarketing experience with frovatriptan:
Among more than 3000 patients with migraine who participated in premarketing clinical trials of FROVA no deaths or serious cardiac events were reported which were related to the use of FROVA.
Cerebrovascular Events and Fatalities with 5-HT1 Agonists:
Cerebral hemorrhage, subarachnoid hemorrhage, stroke and other cerebrovascular events have been reported in patients treated with 5-HT1 agonists; and some have resulted in fatalities. In a number of cases, it appears possible that the cerebrovascular events were primary, the agonist having been administered in the incorrect belief that the symptoms experienced were a consequence of migraine, when they were not. It should be noted that patients with migraine may be at increased risk of certain cerebrovascular events (e.g. stroke, hemorrhage, transient ischemic attack).
Other Vasospasm-Related Events:
5-HT1 agonists may cause vasospastic reactions other than coronary artery spasm. Both peripheral vascular ischemia and colonic ischemia with abdominal pain and bloody diarrhea have been reported with 5-HT1 agonists.
Effects on Blood Pressure:
In young healthy subjects, there were statistically significant increases in systolic and diastolic blood pressure after single doses of 80 mg frovatriptan (32 times the clinical dose) and above. These increases were transient, resolved spontaneously and were not clinically significant. At the recommended dose of 2.5 mg, transient changes in systolic blood pressure were recorded in some elderly subjects (65 - 77 years). Any increases were generally small, resolved spontaneously, and blood pressure remained within the normal range. Frovatriptan is contraindicated in patients with uncontrolled hypertension (see CONTRAINDICATIONS).
An 18% increase in mean pulmonary artery pressure was seen following dosing with another 5-HT1 agonist in a study evaluating subjects undergoing cardiac catheterization.
Serotonin Syndrome:
The development of a potentially life-threatening serotonin syndrome may occur with triptans, including FROVA treatment, particularly during combined use with selective serotonin reuptake inhibitors (SSRIs) or serotonin norepinephrine reuptake inhibitors (SNRIs). If concomitant treatment with FROVA and an SSRI (e.g., fluoxetine, paroxetine, sertraline, fluvoxamine, citalopram, escitalopram) or SNRI (e.g., venlafaxine, duloxetine) is clinically warranted, careful observation of the patient is advised, particularly during treatment initiation and dose increases. Serotonin syndrome symptoms may include mental status changes (e.g., agitation hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination) and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). (See PRECAUTIONS – Drug Interactions).
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
FROVA is indicated for the acute treatment of migraine attacks with or without aura in adults.
FROVA is not intended for the prophylactic therapy of migraine or for use in the management of hemiplegic or basilar migraine (see CONTRAINDICATIONS). The safety and effectiveness of FROVA have not been established for cluster headache, which is present in an older, predominately male, population.
History
There is currently no drug history available for this drug.
Other Information
FROVA (frovatriptan succinate) tablets contain frovatriptan succinate, a selective 5-hydroxy-tryptamine1 (5-HT1B/1D) receptor subtype agonist, as the active ingredient. Frovatriptan succinate is chemically designated as R-(+) 3-methylamino-6-carboxamido-1,2,3,4-tetrahydrocarbazole monosuccinate monohydrate and it has the following structure:
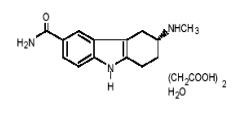
The empirical formula is C14H17N3O.C4H6O4.H2O, representing a molecular weight of 379.4. Frovatriptan succinate is a white to off-white powder that is soluble in water. Each FROVA tablet for oral administration contains 3.91 mg frovatriptan succinate, equivalent to 2.5 mg of frovatriptan base. Each tablet also contains the inactive ingredients lactose NF, microcrystalline cellulose NF, colloidal silicon dioxide NF, sodium starch glycolate NF, magnesium stearate NF, hydroxypropylmethylcellulose USP, polyethylene glycol 3000 USP, triacetin USP, and titanium dioxide USP.
Sources