Methyldopa Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
It is important to recognize that a positive Coombs test, hemolytic anemia, and liver disorders may occur with methyldopa therapy. The rare occurrences of hemolytic anemia or liver disorders could lead to potentially fatal complications unless properly recognized and managed. Read this section carefully to understand these reactions.
With prolonged methyldopa therapy, 10 to 20 percent of patients develop a positive direct Coombs test which usually occurs between 6 and 12 months of methyldopa therapy. Lowest incidence is at daily dosage of 1 g or less. This on rare occasions may be associated with hemolytic anemia, which could lead to potentially fatal complications. One cannot predict which patients with a positive direct Coombs test may develop hemolytic anemia.
Prior existence or development of a positive direct Coombs test is not in itself a contraindication to use of methyldopa. If a positive Coombs test develops during methyldopa therapy, the physician should determine whether hemolytic anemia exists and whether the positive Coombs test may be a problem. For example, in addition to a positive direct Coombs test there is less often a positive indirect Coombs test which may interfere with cross matching of blood.
Before treatment is started, it is desirable to do a blood count (hematocrit, hemoglobin, or red cell count) for a baseline or to establish whether there is anemia. Periodic blood counts should be done during therapy to detect hemolytic anemia. It may be useful to do a direct Coombs test before therapy and at 6 and 12 months after the start of therapy.
If Coombs-positive hemolytic anemia occurs, the cause may be methyldopa and the drug should be discontinued. Usually the anemia remits promptly. If not, corticosteroids may be given and other causes of anemia should be considered. If the hemolytic anemia is related to methyldopa, the drug should not be reinstituted.
When methyldopa causes Coombs positivity alone or with hemolytic anemia, the red cell is usually coated with gamma globulin of the IgG (gamma G) class only. The positive Coombs test may not revert to normal until weeks to months after methyldopa is stopped.
Should the need for transfusion arise in a patient receiving methyldopa, both a direct and an indirect Coombs test should be performed. In the absence of hemolytic anemia, usually only the direct Coombs test will be positive. A positive direct Coombs test alone will not interfere with typing or cross matching. If the indirect Coombs test is also positive, problems may arise in the major cross match and the assistance of a hematologist or transfusion expert will be needed.
Occasionally, fever has occurred within the first 3 weeks of methyldopa therapy, associated in some cases with eosinophilia or abnormalities in one or more liver function tests, such as serum alkaline phosphatase, serum transaminases (SGOT, SGPT), bilirubin and prothrombin time. Jaundice, with or without fever, may occur with onset, usually within the first 2 or 3 months of therapy. In some patients the findings are consistent with those of cholestasis. In others the findings are consistent with hepatitis and hepatocellular injury.
Rarely, fatal hepatic necrosis has been reported after use of methyldopa. These hepatic changes may represent hypersensitivity reactions. Periodic determinations of hepatic function should be done, particularly during the first 6 to 12 weeks of therapy or whenever an unexplained fever occurs. If fever, abnormalities in liver function tests, or jaundice appear, stop therapy with methyldopa. If caused by methyldopa, the temperature and abnormalities in liver function characteristically have reverted to normal when the drug was discontinued. Methyldopa should not be reinstituted in such patients.
Rarely, a reversible reduction of the white blood cell count with a primary effect on the granulocytes has been seen. The granulocyte count returned promptly to normal on discontinuance of the drug. Rare cases of granulocytopenia have been reported. In each instance, upon stopping the drug, the white cell count returned to normal. Reversible thrombocytopenia has occurred rarely.
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
Hypertension.
History
There is currently no drug history available for this drug.
Other Information
Methyldopa USP is an antihypertensive drug.
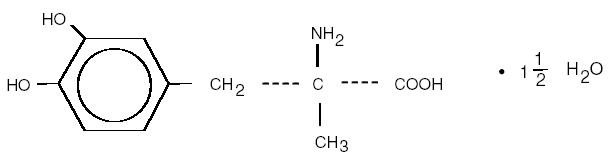
Methyldopa USP is the L-isomer of alpha-methyldopa. Its chemical name is levo-3-(3,4-dihydroxyphenyl)- 2-methylalanine sesquihydrate. Its structural formula is:
C 10H13NO4 • 1 1/2 H2O M.W. 238.24
Methyldopa USP is a white to yellowish white, odorless fine powder and is sparingly soluble in water.
Each tablet, for oral administration, contains 250 mg or 500 mg of methyldopa USP. Potency is calculated in the anhydrous basis. Inactive ingredients: citric acid, colloidal silicon dioxide, edetate disodium, ethylcellulose, hypromellose, magnesium stearate, methylcellulose, polyethylene glycol, sodium starch glycolate, and titanium dioxide. In addition, the 250 mg tablet contains calcium sulfate, hydroxypropyl cellulose, and talc, and the 500 mg tablet contains polysorbate 80.
Sources



![Methyldopa Tablet, Film Coated [Rebel Distributors Corp]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=60a57abf-9487-4093-9709-40f05755e7b1&name=cf051ddf-aca5-4bdc-ac16-27310d5fd59a-02.jpg)
![Methyldopa Tablet, Film Coated [Accord Healthcare Inc.]](http://www.recallguide.org/wp-content/themes/bootstrap/assets/img/drug-image-placeholder.jpg)
![Methyldopa Tablet [Mylan Institutional Inc.]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=57c496b3-10ab-4a72-8d8f-42a2256189dd&name=isopropyl-alcohol-70-percent_ric-1.jpg)
![Methyldopa Tablet [Remedyrepack Inc. ]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=4eb98b20-561d-453d-8f64-88d1e60faff6&name=MM2.jpg)
![Methyldopa Tablet, Film Coated [Rebel Distributors Corp]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=fa1c61c2-7e24-40ff-916e-456b8725a5cd&name=fa1c61c2-7e24-40ff-916e-456b8725a5cd-02.jpg)
![Methyldopa Tablet [Cardinal Health]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=d87d63fd-48f7-4130-af03-5e263c338fe4&name=2a911dc9-a0fa-4efa-8d09-b300038cce28-02.jpg)
![Methyldopa Tablet, Film Coated [Teva Pharmaceuticals Usa Inc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=916d802c-91b6-4015-9e57-7ebf7e9bb5ee&name=2d56136a-9922-4bc6-84ea-d02556511783-02.jpg)
![Methyldopa Tablet, Film Coated [A-s Medication Solutions Llc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=cac2489b-7aa8-4d30-b2e4-2937a65fa7cf&name=0510-0.jpg)
![Methyldopa Tablet [Mylan Pharmaceuticals Inc.]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=3569de40-1642-4771-b5e8-f949cae606f7&name=liquidoxygen.jpg)