FDA records indicate that there are no current recalls for this drug.
Are you a medical professional?
Trending Topics
Prochieve Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
The physician should be alert to the earliest manifestations of thrombotic disorders (thrombophlebitis, cerebrovascular disorders, pulmonary embolism, and retinal thrombosis). Should any of these occur or be suspected, the drug should be discontinued immediately.
Progesterone and progestins have been used to prevent miscarriage in women with a history of recurrent spontaneous pregnancy losses. No adequate evidence is available to show that they are effective for this purpose.
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
Prochieve® 8% is indicated for progesterone supplementation or replacement as part of an Assisted Reproductive Technology ("ART") treatment for infertile women with progesterone deficiency.
Prochieve® 4% is indicated for the treatment of secondary amenorrhea. Prochieve® 8% is indicated for use in women who have failed to respond to treatment with Prochieve® 4%.
History
There is currently no drug history available for this drug.
Other Information
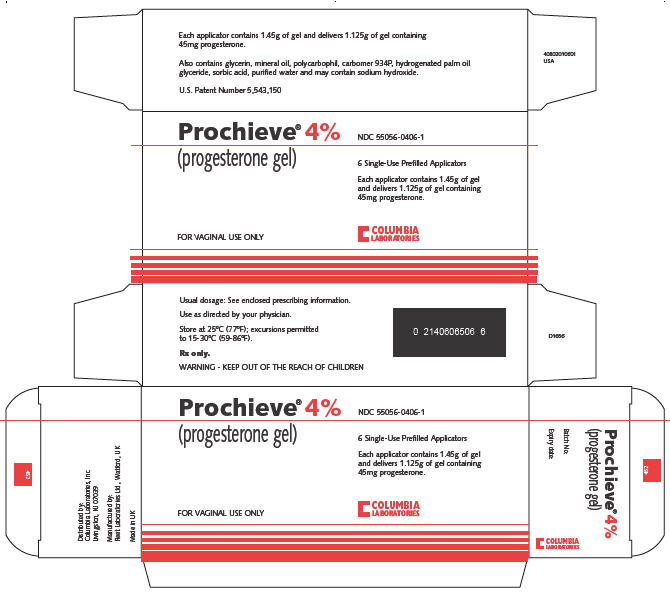
Prochieve® (progesterone gel) is a bioadhesive vaginal gel containing micronized progesterone in an emulsion system, which is contained in single use, one piece polyethylene vaginal applicators. The carrier vehicle is an oil in water emulsion containing the water swellable, but insoluble polymer, polycarbophil. The progesterone is partially soluble in both the oil and water phase of the vehicle, with the majority of the progesterone existing as a suspension. Physically, Prochieve® has the appearance of a soft, white to off-white gel.
The active ingredient, progesterone, is present in either a 4% or an 8% concentration (w/w). The chemical name for progesterone is pregn-4-ene-3,20-dione. It has an empirical formula of C21H30O2 and a molecular weight of 314.5.
The structural formula is:

Progesterone exists in two polymorphic forms. Form 1, which is the form used in Prochieve®, exists as white orthorhombic prisms with a melting point of 127-131°C.
Each applicator delivers 1.125 grams of Prochieve® gel containing either 45 mg (4% gel) or 90 mg (8% gel) of progesterone in a base containing glycerin, mineral oil, polycarbophil, carbomer 934P, hydrogenated palm oil glyceride, sorbic acid, purified water and may contain sodium hydroxide.
Sources
Prochieve Manufacturers
-
Columbia Laboratories
![Prochieve (Progesterone) Gel [Columbia Laboratories]](/wp-content/themes/bootstrap/assets/img/loading2.gif)
Prochieve | Columbia Laboratories
![Prochieve (Progesterone) Gel [Columbia Laboratories] Prochieve (Progesterone) Gel [Columbia Laboratories]](/wp-content/themes/bootstrap/assets/img/loading2.gif)
Assisted Reproductive TechnologyProchieve® 8% is administered vaginally at a dose of 90 mg once daily in women who require progesterone supplementation. Prochieve® 8% is administered vaginally at a dose of 90 mg twice daily in women with partial or complete ovarian failure who require progesterone replacement. If pregnancy occurs, treatment may be continued until placental autonomy is achieved, up to 10-12 weeks.
Secondary AmenorrheaProchieve® 4% is administered vaginally every other day up to a total of six doses. For women who fail to respond, a trial of Prochieve® 8% every other day up to a total of six doses may be instituted.
It is important to note that a dosage increase from the 4% gel can only be accomplished by using the 8% gel. Increasing the volume of gel administered does not increase the amount of progesterone absorbed.
SEE Prochieve® PATIENT INFORMATION SHEET - HOW TO USE Prochieve®.
Note: The PATIENT INFORMATION SHEET contains special instructions for using the applicator at altitudes above 2500 feet in order to avoid a partial release of Prochieve® before vaginal insertion.
Login To Your Free Account