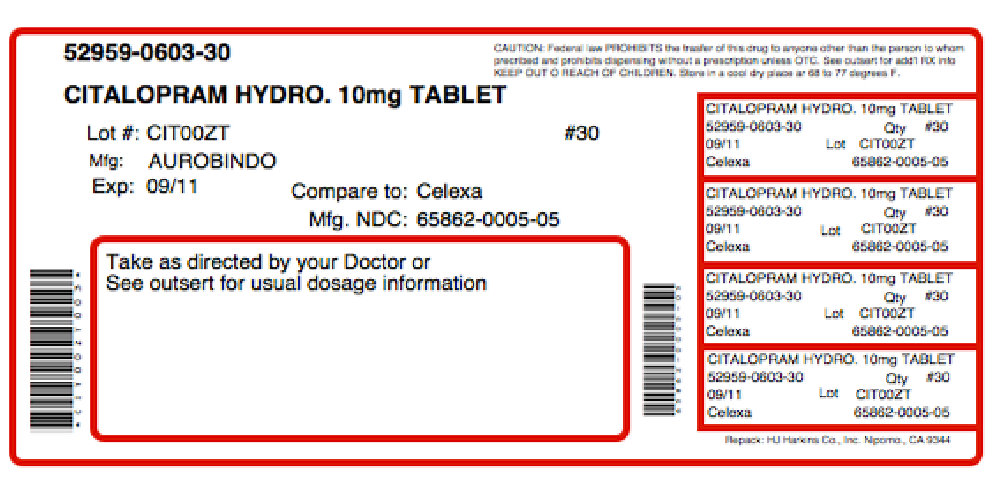
FDA records indicate that there are no current recalls for this drug.
Are you a medical professional?
Trending Topics
Sentralopram Am-10 Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
WARNINGS
Clinical Worsening and Suicide Risk
Patients with major depressive disorder (MDD), both adult and pediatric, may experience worsening of their depression and/or the emergence of suicidal ideation and behavior (suicidality) or unusual changes in behavior, whether or not they are taking antidepressant medications, and this risk may persist until significant remission occurs. Suicide is a known risk of depression and certain other psychiatric disorders, and these disorders themselves are the best predictors of suicide. There has been a long-standing concern, however, that antidepressants may have a role in inducing worsening of depression and the emergence of suicidality in certain patients during the early phases of treatment.
Pooled analyses of short-term placebo-controlled trials of antidepressant drugs (SSRIs and others) showed that these drugs increase the risk of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults (ages 18-24) with major depressive disorder (MDD) and other psychiatric disorders. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction with antidepressants compared to placebo in adults aged 65 and older.
The pooled analyses of placebo-controlled trials in children and adolescents with MDD, obsessive compulsive disorder (OCD), or other psychiatric disorders included a total of 24 short-term trials of 9 antidepressant drugs in over 4400 patients. The pooled analyses of placebo-controlled trials in adults with MDD or other psychiatric disorders included a total of 295 short-term trials (median duration of 2 months) of 11 antidepressant drugs in over 77,000 patients. There was considerable variation in risk of suicidality among drugs, but a tendency toward an increase in the younger patients for almost all drugs studied. There were differences in absolute risk of suicidality across the different indications, with the highest incidence in MDD. The risk differences (drug vs. placebo), however, were relatively stable within age strata and across indications. These risk differences (drug-placebo difference in the number of cases of suicidality per 1000 patients treated) are provided in Table 1.
| Age Range | Drug-Placebo Difference in Number of Cases of Suicidality per 1000 Patients Treated |
| |
Increases Compared to Placebo |
| less than 18 | 14 additional cases |
| 18-24 | 5 additional cases |
| |
Decreases Compared to Placebo |
| 25-64 | 1 fewer case |
| greater than or equal to 65 | 6 fewer cases |
No suicides occurred in any of the pediatric trials. There were suicides in the adult trials, but the number was not sufficient to reach any conclusion about drug effect on suicide.
It is unknown whether the suicidality risk extends to longer-term use, i.e., beyond several months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with depression that the use of antidepressants can delay the recurrence of depression.
All patients being treated with antidepressants for any indication should be monitored appropriately and observed closely for clinical worsening, suicidality, and unusual changes in behavior, especially during the initial few months of a course of drug therapy, or at times of dose changes, either increases or decreases.
The following symptoms, anxiety, agitation, panic attacks, insomnia, irritability, hostility, aggressiveness, impulsivity, akathisia (psychomotor restlessness), hypomania, and mania, have been reported in adult and pediatric patients being treated with antidepressants for major depressive disorder as well as for other indications, both psychiatric and nonpsychiatric. Although a causal link between the emergence of such symptoms and either the worsening of depression and/or the emergence of suicidal impulses has not been established, there is concern that such symptoms may represent precursors to emerging suicidality.
Consideration should be given to changing the therapeutic regimen, including possibly discontinuing the medication, in patients whose depression is persistently worse, or who are experiencing emergent suicidality or symptoms that might be precursors to worsening depression or suicidality, especially if these symptoms are severe, abrupt in onset, or were not part of the patient's presenting symptoms.
If the decision has been made to discontinue treatment, medication should be tapered, as rapidly as is feasible, but with recognition that abrupt discontinuation can be associated with certain symptoms (see PRECAUTIONS and DOSAGE AND ADMINISTRATION—Discontinuation of Treatment with Citalopram Tablets, for a description of the risks of discontinuation of citalopram tablets).
Families and caregivers of patients being treated with antidepressants for major depressive disorder or other indications, both psychiatric and nonpsychiatric, should be alerted about the need to monitor patients for the emergence of agitation, irritability, unusual changes in behavior, and the other symptoms described above, as well as the emergence of suicidality, and to report such symptoms immediately to health care providers.Such monitoring should include daily observation by families and caregivers. Prescriptions for citalopram should be written for the smallest quantity of tablets consistent with good patient management, in order to reduce the risk of overdose.
Screening Patients for Bipolar Disorder
A major depressive episode may be the initial presentation of bipolar disorder. It is generally believed (though not established in controlled trials) that treating such an episode with an antidepressant alone may increase the likelihood of precipitation of a mixed/manic episode in patients at risk for bipolar disorder. Whether any of the symptoms described above represent such a conversion is unknown. However, prior to initiating treatment with an antidepressant, patients with depressive symptoms should be adequately screened to determine if they are at risk for bipolar disorder; such screening should include a detailed psychiatric history, including a family history of suicide, bipolar disorder, and depression. It should be noted that citalopram is not approved for use in treating bipolar depression.
Potential for Interaction with Monoamine Oxidase Inhibitors
In patients receiving serotonin reuptake inhibitor drugs in combination with a monoamine oxidase inhibitor (MAOI), there have been reports of serious, sometimes fatal, reactions including hyperthermia, rigidity, myoclonus, autonomic instability with possible rapid fluctuations of vital signs, and mental status changes that include extreme agitation progressing to delirium and coma. These reactions have also been reported in patients who have recently discontinued SSRI treatment and have been started on an MAOI. Some cases presented with features resembling neuroleptic malignant syndrome. Furthermore, limited animal data on the effects of combined use of SSRIs and MAOIs suggest that these drugs may act synergistically to elevate blood pressure and evoke behavioral excitation. Therefore, it is recommended that citalopram tablets should not be used in combination with an MAOI, or within 14 days of discontinuing treatment with an MAOI. Similarly, at least 14 days should be allowed after stopping citalopram tablets before starting an MAOI.
Serotonin Syndrome or Neuroleptic Malignant Syndrome (NMS)-like Reactions
The development of a potentially life-threatening serotonin syndrome or Neuroleptic Malignant Syndrome (NMS)-like reactions have been reported with SNRIs and SSRIs alone, including citalopram treatment, but particularly with concomitant use of serotonergic drugs (including triptans) with drugs which impair metabolism of serotonin (including MAOIs), or with antipsychotics or other dopamine antagonists. Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g., hyperreflexia, incoordination) and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Serotonin syndrome, in its most severe form can resemble neuroleptic malignant syndrome, which includes hyperthermia, muscle rigidity, autonomic instability with possible rapid fluctuation of vital signs, and mental status changes. Patients should be monitored for the emergence of serotonin syndrome or NMS-like signs and symptoms.
The concomitant use of citalopram with MAOIs intended to treat depression is contraindicated. If concomitant treatment of citalopram with a 5-hydroxytryptamine receptor agonist (triptan) is clinically warranted, careful observation of the patient is advised, particularly during treatment initiation and dose increases.
The concomitant use of citalopram with serotonin precursors (such as tryptophan) is not recommended. Treatment with citalopram and any concomitant serotonergic or antidopaminergic agents, including antipsychotics, should be discontinued immediately if the above events occur and supportive symptomatic treatment should be initiated.
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
INDICATIONS AND USAGE
Citalopram tablets are indicated for the treatment of depression.
The efficacy of citalopram tablets in the treatment of depression was established in 4-6 week, controlled trials of outpatients whose diagnosis corresponded most closely to the DSM-III and DSM-III-R category of major depressive disorder (see CLINICAL PHARMACOLOGY).
A major depressive episode (DSM-IV) implies a prominent and relatively persistent (nearly every day for at least 2 weeks) depressed or dysphoric mood that usually interferes with daily functioning, and includes at least five of the following nine symptoms: depressed mood, loss of interest in usual activities, significant change in weight and/or appetite, insomnia or hypersomnia, psychomotor agitation or retardation, increased fatigue, feelings of guilt or worthlessness, slowed thinking or impaired concentration, a suicide attempt or suicidal ideation.
The antidepressant action of citalopram tablets in hospitalized depressed patients has not been adequately studied.
The efficacy of citalopram tablets in maintaining an antidepressant response for up to 24 weeks following 6 to 8 weeks of acute treatment was demonstrated in two placebo-controlled trials (see CLINICAL PHARMACOLOGY). Nevertheless, the physician who elects to use citalopram tablets for extended periods should periodically re-evaluate the long-term usefulness of the drug for the individual patient.
INDICATIONS FOR USE Sentra AM is intended for the clinical nutritional management of the metabolic processes associated with fatigue and cognitive disorders. - Chronic fatigue - Cognitive impairment - Fibromyalgia
History
There is currently no drug history available for this drug.
Other Information
DESCRIPTION
Citalopram hydrobromide is an orally administered selective serotonin reuptake inhibitor (SSRI) with a chemical structure unrelated to that of other SSRIs or of tricyclic, tetracyclic, or other available antidepressant agents. Citalopram hydrobromide is a racemic bicyclic phthalane derivative designated (±)-1-(3-dimethylaminopropyl)-1-(4-fluorophenyl)-1,3-dihydroisobenzofuran-5-carbonitrile, hydrobromide with the following structural formula:

The molecular formula is C20H22BrFN2O and its molecular weight is 405.35.
Citalopram hydrobromide occurs as a fine, white to off-white powder. Citalopram hydrobromide is sparingly soluble in water and soluble in ethanol.
Citalopram hydrobromide is available as tablets.
Citalopram 10 mg tablets are biconvex, round shaped film coated tablets in strengths equivalent to 10 mg citalopram base. Citalopram 20 mg and 40 mg tablets are scored biconvex capsule shaped film coated tablets containing citalopram hydrobromide in strengths equivalent to 20 mg or 40 mg citalopram base. The tablets also contain the following inactive ingredients: copovidone, corn starch, croscarmellose sodium, lactose monohydrate, magnesium stearate, hypromellose, microcrystalline cellulose, polyethylene glycol, and titanium dioxide. Iron oxides are used as coloring agents in the peach (10 mg) and light pink (20 mg) tablets.
PRODUCT DESCRIPTION Primary Ingredients Sentra AM consists of a proprietary blend of amino acids, cocoa, and flavonoids in specific proportions. These ingredients fall into the category of “Generally Regarded as Safe” (GRAS) as defined by the Food and Drug Administration (FDA) (Sections 201(s) and 409 of the Federal Food, Drug, and Cosmetic Act). A GRAS substance is distinguished from a food additive on the basis of the common knowledge about the safety of the substance for its intended use. The standard for an ingredient to achieve GRAS status requires not only technical demonstration of non-toxicity and safety, but also general recognition of safety through widespread usage and agreement of that safety by experts in the field. Many ingredients have been determined by the U.S. Food and Drug Administration (FDA) to be GRAS, and are listed as such by regulation, in Volume 21 Code of Federal Regulations (CFR) Sections 182, 184, and 186. Amino Acids Amino Acids are the building blocks of protein. All amino acids are GRAS listed as they have been ingested by humans for thousands of years. The doses of the amino acids, particularly choline, in Sentra AM are equivalent to those found in the usual human diet; however the formulation uses specific ratios of the key ingredients to elicit a therapeutic response. Patients with fatigue and cognitive disorders may require an increased amount of certain amino acids that cannot be obtained from normal diet alone. Choline, for example, is an obligatory amino acid. The body cannot make choline and must obtain choline from the diet. Choline is needed to produce acetylcholine. Acetylcholine is required to reduce fatigue and improve cognitive function. Patients with fatigue and cognitive disorders have altered choline metabolism. Some patients with fatigue and cognitive disorders have a resistance to the metabolism of choline that is similar to the mechanism found in insulin resistance. Patients with fatigue and cognitive disorders cannot acquire sufficient choline from the diet without ingesting a prohibitively large amount of calories, particularly calories from protein. Flavonoids Flavonoids are a group of phytochemical compounds found in all vascular plants including fruits and vegetables. They are a part of a larger class of compounds known as polyphenols. Many of the therapeutic or health benefits of colored fruits and vegetables, cocoa, red wine, and green tea are directly related to their flavonoid content. The specially formulated flavonoids found in Sentra AM cannot be obtained from conventional foods in the necessary proportions to elicit a therapeutic response. Physical Description Sentra AM is a yellow to light brown powder. Sentra AM contains L-Glutamic Acid, Choline Bitartrate, Cocoa, Acetylcarnitine, and Hawthorn Berry. Other Ingredients Sentra AM contains the following inactive or other ingredients as fillers, excipients, and colorings: magnesium stearate, microcrystalline cellulose, Maltodextrin NF, gelatin (as the capsule material).
Sources
Sentralopram Am-10 Manufacturers
-
Physician Therapeutics Llc
![Sentralopram Am-10 (Citalopram Hydrobromide, Choline) Kit [Physician Therapeutics Llc]](/wp-content/themes/bootstrap/assets/img/loading2.gif)
Sentralopram Am-10 | Physician Therapeutics Llc
![Sentralopram Am-10 (Citalopram Hydrobromide, Choline) Kit [Physician Therapeutics Llc] Sentralopram Am-10 (Citalopram Hydrobromide, Choline) Kit [Physician Therapeutics Llc]](/wp-content/themes/bootstrap/assets/img/loading2.gif)
DOSAGE AND ADMINISTRATION
Initial Treatment
Citalopram tablets should be administered at an initial dose of 20 mg once daily, generally with an increase to a dose of 40 mg/day. Dose increases should usually occur in increments of 20 mg at intervals of no less than one week. Although certain patients may require a dose of 60 mg/day, the only study pertinent to dose response for effectiveness did not demonstrate an advantage for the 60 mg/day dose over the 40 mg/day dose; doses above 40 mg are therefore not ordinarily recommended.
Citalopram tablets should be administered once daily, in the morning or evening, with or without food.
Special Populations
20 mg/day is the recommended dose for most elderly patients and patients with hepatic impairment, with titration to 40 mg/day only for nonresponding patients.
No dosage adjustment is necessary for patients with mild or moderate renal impairment. Citalopram tablets should be used with caution in patients with severe renal impairment.
Treatment of Pregnant Women During the Third Trimester
Neonates exposed to citalopram and other SSRIs or SNRIs, late in the third trimester, have developed complications requiring prolonged hospitalization, respiratory support, and tube feeding (see PRECAUTIONS). When treating pregnant women with citalopram during the third trimester, the physician should carefully consider the potential risks and benefits of treatment. The physician may consider tapering citalopram in the third trimester.
Maintenance Treatment
It is generally agreed that acute episodes of depression require several months or longer of sustained pharmacologic therapy. Systematic evaluation of citalopram tablets in two studies has shown that its antidepressant efficacy is maintained for periods of up to 24 weeks following 6 or 8 weeks of initial treatment (32 weeks total). In one study, patients were assigned randomly to placebo or to the same dose of citalopram tablets (20-60 mg/day) during maintenance treatment as they had received during the acute stabilization phase, while in the other study, patients were assigned randomly to continuation of citalopram tablets 20 or 40 mg/day, or placebo, for maintenance treatment. In the latter study, the rates of relapse to depression were similar for the two dose groups (see Clinical Trials under CLINICAL PHARMACOLOGY). Based on these limited data, it is not known whether the dose of citalopram needed to maintain euthymia is identical to the dose needed to induce remission. If adverse reactions are bothersome, a decrease in dose to 20 mg/day can be considered.
Discontinuation of Treatment with Citalopram Tablets
Symptoms associated with discontinuation of citalopram and other SSRIs and SNRIs have been reported (see PRECAUTIONS). Patients should be monitored for these symptoms when discontinuing treatment. A gradual reduction in the dose rather than abrupt cessation is recommended whenever possible. If intolerable symptoms occur following a decrease in the dose or upon discontinuation of treatment, then resuming the previously prescribed dose may be considered. Subsequently, the physician may continue decreasing the dose but at a more gradual rate.
Switching Patients To or From a Monoamine Oxidase Inhibitor
At least 14 days should elapse between discontinuation of an MAOI and initiation of citalopram tablets therapy. Similarly, at least 14 days should be allowed after stopping citalopram tablets before starting an MAOI (see CONTRAINDICATIONS and WARNINGS).DOSAGE AND ADMINISTRATION Recommended Administration For the nutritional management of the metabolic processes in patients with fatigue and cognitive disorders. Take (2) capsules one to three times daily or as directed by physician. As with most amino acid formulations Sentra AM should be taken between meals without food to increase the absorption of key ingredients.
Login To Your Free Account