Uroxatral Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
There is currently no warning information available for this product. We apologize for any inconvenience.
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
1 INDICATIONS AND USAGE
UROXATRAL is indicated for the treatment of signs and symptoms of benign prostatic hyperplasia. UROXATRAL is not indicated for the treatment of hypertension.
2 DOSAGE AND ADMINISTRATION
The recommended dosage is one 10 mg UROXATRAL (alfuzosin HCl) extended-release tablet once daily. The extent of absorption of Uroxatral is 50% lower under fasting conditions. Therefore, Uroxatral should be taken immediately after the same meal each day. The tablets should not be chewed or crushed.
3 DOSAGE FORMS AND STRENGTHS
UROXATRAL (alfuzosin HCl) extended-release tablet 10 mg is available as a round, three-layer tablet: one white layer between two yellow layers, debossed with X10.
4 CONTRAINDICATIONS
UROXATRAL is contraindicated for use in patients with moderate or severe hepatic impairment (Childs-Pugh categories B and C), since alfuzosin blood levels are increased in these patients. [see Clinical Pharmacology (12.3)].
UROXATRAL is contraindicated for use with potent CYP3A4 inhibitors such as ketoconazole, itraconazole, and ritonavir, since alfuzosin blood levels are increased. [see Clinical Pharmacology (12.3)].
UROXATRAL is contraindicated in patients known to be hypersensitive to alfuzosin hydrochloride or any component of UROXATRAL tablets.
History
There is currently no drug history available for this drug.
Other Information
11 DESCRIPTION
Each UROXATRAL extended-release tablet contains 10 mg alfuzosin hydrochloride as the active ingredient. Alfuzosin hydrochloride is a white to off-white crystalline powder that melts at approximately 240°C. It is freely soluble in water, sparingly soluble in alcohol, and practically insoluble in dichloromethane.
Alfuzosin hydrochloride is (R,S)-N-[3-[(4-amino-6,7-dimethoxy-2-quinazolinyl) methylamino] propyl] tetrahydro-2-furancarboxamide hydrochloride. The empirical formula of alfuzosin hydrochloride is C19H27N5O4•HCl. The molecular weight of alfuzosin hydrochloride is 425.9. Its structural formula is:
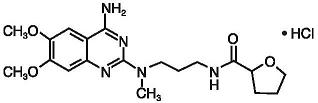
The tablet also contains the following inactive ingredients: colloidal silicon dioxide (NF), ethylcellulose (NF), hydrogenated castor oil (NF), hydroxypropyl methylcellulose (USP), magnesium stearate (NF), mannitol (USP), microcrystalline cellulose (NF), povidone (USP), and yellow ferric oxide (NF).
Sources